| Issue |
Eur. Phys. J. Appl. Phys.
Volume 24, Number 1, October 2003
|
|
|---|---|---|
| Page(s) | 33 - 36 | |
| Section | Imaging, Microscopy and Spectroscopy | |
| DOI | https://doi.org/10.1051/epjap:2003052 | |
| Published online | 16 July 2003 | |
https://doi.org/10.1051/epjap:2003052
Calorimetric studies of bulk Se-Te-Pb glassy system
Semiconductors Laboratory, Department of Applied Physics,
Guru Nanak Dev University, Amritsar, 143005, India
Corresponding author: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
19
November
2002
Revised:
12
May
2003
Accepted:
2
June
2003
Published online:
16
July
2003
Abstract
Calorimetric studies have been performed on the Se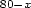 Te20 Pbx
(
Te20 Pbx
(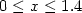 ) samples at
different heating rates (5−20 °C/min). The glass transition temperature Tg, the peak
crystallization temperature Tp, and the melting temperature Tm are found to increase with
increase in heating rate. The apparent activation energies for glass transition Et and for
crystallization Ec have been calculated from the heating rate dependence of Tg and Tp
respectively. The effect of Pb addition to the Se-Te matrix on the mechanism of crystal
growth has been investigated. The glass transition temperature Tg and the peak
crystallization temperature Tp initially decrease with small addition of Pb (x = 0.6) but increases with further addition of Pb (x > 0.6). The thermal stability and glass-forming
tendency have also been studied.
) samples at
different heating rates (5−20 °C/min). The glass transition temperature Tg, the peak
crystallization temperature Tp, and the melting temperature Tm are found to increase with
increase in heating rate. The apparent activation energies for glass transition Et and for
crystallization Ec have been calculated from the heating rate dependence of Tg and Tp
respectively. The effect of Pb addition to the Se-Te matrix on the mechanism of crystal
growth has been investigated. The glass transition temperature Tg and the peak
crystallization temperature Tp initially decrease with small addition of Pb (x = 0.6) but increases with further addition of Pb (x > 0.6). The thermal stability and glass-forming
tendency have also been studied.
PACS: 77.84.Bw – Elements, oxides, nitrides, borides, carbides, chalcogenides, etc. / 64.70.Pf – Glass transitions / 68.60.Dv – Thermal stability; thermal effects
© EDP Sciences, 2003
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


