| Issue |
Eur. Phys. J. Appl. Phys.
Volume 86, Number 2, May 2019
Materials for energy harvesting, conversion, storage and environmental engineering (Icome 2018)
|
|
|---|---|---|
| Article Number | 20201 | |
| Number of page(s) | 4 | |
| Section | Physics of Organic Materials and Devices | |
| DOI | https://doi.org/10.1051/epjap/2019190047 | |
| Published online | 10 June 2019 | |
https://doi.org/10.1051/epjap/2019190047
Regular Article
Open circuit voltage of organic photovoltaic cells using C60 as acceptor: variation with the donor★
1
Institut des Matériaux Jean Rouxel (IMN), CNRS, UMR 6502, Université de Nantes, 2 Rue de la Houssinière, BP 32229, 44322 Nantes Cedex 3, France
2
Equipe de Recherche en Couches Minces et Nanomatériaux (ERCMN), Faculté des Sciences et Techniques, Université Abdelmalek Essadi, Tanger, Maroc
3
MOLTECH-Anjou, CNRS, UMR 6200, Université de Nantes, 2 Rue de la Houssinière, BP 92208, Nantes 44000, France
* e-mail: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
26
February
2019
Received in final form:
4
April
2019
Accepted:
15
April
2019
Published online: 10 June 2019
Abstract
The open circuit voltage (Voc) of organic photovoltaic cells (OPVs) is an important parameter in terms of OPV performance. In the present work, we check that its value depends on the energy difference between the Lowest Unoccupied Molecular Orbital of the electron acceptor (LUMOA) and the Highest Occupied Molecular Orbital of the donor (HOMOD). The electron acceptor is the fullerene, while the electron acceptors are used as parameter. The results show that Voc increases with the value of Δ(LUMOA–HOMOD). However, for some molecules, this increase is not linear, which shows that other parameters are also determinant.
Contribution to the Topical Issue “Materials for energy harvesting, conversion, storage and environmental engineering (Icome 2018)”, edited by Jean-Michel Nunzi, Rachid Bennacer, Mohammed El Ganaoui, Mohamed El Jouad
© Z. El Jouad et al., EDP Sciences, 2019
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
1 Introduction
Today, it appears that the different PVC families are not exclusive but complementary, crystalline Si (C-Si) and inorganic thin films solar cells (GaAs, CuInSe2, etc.) for their high efficiency and stability, perovskite (PVSCs) for its high potential in the near future and organic solar cells (OPVs) for their high potential in the field of semitransparent OPVs.
In the case of organic materials, a specific interest is dedicated to flexible semitransparent OPVs, the aim being integrating OPVs into façade elements to reduce the energy consumption and CO2 emission of a building. Actually, beside their flexible, lightweight, the most exciting thing about organic semiconductors is that it is possible to design molecules that are colored or quite transparent, which allows growing semitransparent photovoltaic cells. Concerning the pronounced absorption features of various absorber molecules used in organic solar cells, devices can be designed to transmit light in a specific spectral range. Thereby, colored see-through photovoltaic elements (e.g., windows or colored semitransparent curtains) are possible [1]. If some organic molecules are sensitive to humidity and oxygen, it was already shown that an efficient encapsulation allows over passing this problem and a life times of more than 8 yr were already announced. OPVs are already on the market as semitransparent solar cells into building façades [1] and also as mobile phone chargers [2].
Even if the efficiency of OPVs progresses regularly it is always lower than that of inorganic cells. The main parameters on which the efficiency depends are the short circuit current, J sc, the fill factor, FF, and the open circuit voltage, Voc. The OPVs are based on the organic couple electron donor/electron acceptor, (ED)/(EA) [3]. In the present work, we studied the behavior of Voc when we change the electron donor. It is now well admitted that the Voc maximum value is limited by the difference between the Lowest Unoccupied Molecular Orbital of the acceptor (LUMOA) and the Highest Occupied Molecular Orbital of the donor (HOMOD) [4] (Fig. 1). If Voc increases with Δ(LUMOA–HOMOD), the light absorption efficiency depends on the band gap value, Eg. The optimum band gap value is: 1.5 eV < Eg < 2 eV. Moreover, the energy difference between the LUMOs (and HOMOs) of the donor and acceptor must be higher than the exciton energy in order to achieve efficient charge separation. All this makes it necessary to find the optimum compromise, to achieve the highest possible efficiency (Fig. 1). In the present study, in order to obtain reproducible experimental conditions, we choose to work on planar-heterojunctions. It was already shown that efficiency as high as 8.4% can be achieved using small molecules [5]. Actually, if, often, OPVs based on bulk-heterojunction (BHJ) achieve highest efficiency, their morphology is difficult to control. Therefore, we used simple stacked layers sandwiched between the ITO anode and the Al cathode [3]. We used fullerene as acceptor, while we used the electron donor as parameter. We show that Voc increases with Δ(LUMOA–HOMOD), the slope of the corresponding curve being around 1.
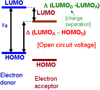 |
Fig. 1 Band structure of the interface donor/acceptor. |
2 Experimental technique
2.1 Organic photovoltaic cells (OPVs) realization
ITO coated glass substrates with a sheet resistance of about 25 Ω/square were used as anode. After cleaning and drying, the substrates were introduced into a vacuum deposition apparatus. All the layers were deposited by Joule effect without breaking the vacuum. The thickness and the deposition rate of the layers were controlled by a quartz monitor. The hole transporting layer (HTL)and the electron transporting layer (ETL) of all the OPVs were MoO3/CuI and Alq3, respectively [6]. The deposition rate of the organic layer was 0.05 nm/s while their thickness was optimized for each ED/EA couple. Fullerene was used as EA, while different EDs were probed. Al was used as cathode, the active surface of the OPVs was 0.10 cm2. Finally the OPVs were as follow: ITO/MoO3 (3 nm)/CuI (1.5 nm)/ED (x nm)/C60 (40 nm)/Alq3 (9 nm)/Al (120 nm).
2.2 Organic photovoltaic cells (OPVs) characterizations
The electrical characterization of the OPVs was performed using a J–V tester, in the dark and under sun global AM 1.5 simulated solar illumination. The ED/EA bilayers were also characterized by X-ray diffraction and optical absorption. The bilayer structures were analyzed by X-ray diffraction (XRD) by a Siemens D5000 diffractometer using Kα radiation from Cu (λ Kα = 0.15406 nm). Optical absorption spectra were recorded on a Carry spectrophotometer from 300 nm to 900 nm. Also, some images of the surfaces were obtained by Atomic Force Microscopy (AFM).
3 Experimental results and discussion
In order to check the variation of Voc with LOMOD, the LUMOA being fixed (LUMOC60 = 4.4 eV), we used ED with different HOMO values. These ED were either commercial (Phthalocyanine dyes [7], Pentacene [8]; Scheme 1) or synthesized in laboratories (Pentathiophene [8], DBP [9], M8-1 [10], M8-2 [10], BSTV [11]; Scheme 2). Small molecules used in this work are known for their harmless nature. For instance, Phthalocyanine dyes, such as CuPc, are used in gouache paint for children [12].
The HOMOD and the corresponding Δ(LUMOA–HOMOD), are given in Table 1. It can be seen that a quite wide range of HOMOD (from 4.9 eV for pentacene to 5.65 eV for BSTV) and of Δ(LUMOA–HOMOD) (from 0.5 eV to 1.52 eV) were probed. The experimental values of Voc are also reported in Table 1. It must be noted that the highest efficiency was obtained with the couple SubPc/C60 (Voc = 1.03 V; J SC = 8.31; FF = 54.5% and η = 4.66%), while the worst result is obtained with the couple pentacene/C60 (Voc = 0.35 V; J SC = 3.74; FF = 40% and η = 0.52%) (Fig. 2).
It can be seen immediately in Table 1 that the value of Voc has a determining effect on the efficiency of the cell since the highest efficiency corresponds to the largest Voc and the lowest to the smallest Voc.
As expected, Voc increases with the value of Δ(LUMOA–HOMOD). Nevertheless, as shown in Figure 3, some scattering is observed depending on organic material. For instance, if the difference between the maximum theoretical value of Voc, i.e. Δ(LUMOA–HOMOD), and the experimental value can be smaller than 0.2 eV, it can also be higher than 0.5 eV (Fig. 3). We have focused our attention on the two ED/EA couples giving the extreme values in Figure 3, namely, Pentathiophène/C60 for the maximum difference between theoretical and experimental value and SubPc/C60 for the minimum value. Concerning the couple Pentathiophène/C60, Voc is only 0.4 eV, while Δ(LUMOA–HOMOD) is 0.95. Such small Voc can be due the presence of leakage current. The Rs and Rsh values calculated from the J–V curves were Rs = 3 Ω and Rsh = 300 Ω for Pentacene/C60 while they were 4 Ω and 575 Ω, respectively, in the case of SubPc/C60. It was already shown that Voc decreases when Rsh decreases, while the saturation current Is of the diode increases.
Such leakage current can be due to some inhomogeneities of the organic films. Therefore, we proceeded to AFM characterizations. The image of a pentathiophene layer is shown in Figure 4. It can be seen that the layer is quite rough, which can initiate some leakage path. The rms deduced is 18 nm. In relation to this result, the dark current of the diode Pentathiophene/C60 under a polarization of 100 mV is 6.4 10−4 mA/cm2, which is much higher than that obtained with the SubPc/C60 couple which is only 1.4 × 10−4 mA/cm2. Actually, the surface rms of the SubPc layer is only 5 nm [6], which is significantly smaller than that of the pentathiophene layer and can justify the smaller leakage current and therefore the smaller difference between the theoretical and the experimental value of Voc.
Moreover, the reduced dark current obtained with SubPc demonstrates that carrier recombination is weak at the interface SubPc/C60 [13]. All this results in a nearly optimum Voc value.
 |
Scheme 1 Commercial electron donors. |
 |
Scheme 2 Laboratory made ED. |
Parameters and Voc for different electron donors.
 |
Fig. 2 J–V characteristics of OPVs with Pentacene/ C60 and SubPc/C60 active couple. |
 |
Fig. 3 Variation of (Δ(LUMOA–HOMOD) − Voc) with Δ(LUMOA–HOMOD) |
 |
Fig. 4 3D image of a pentathiophene layer with thickness 70 nm. |
4 Conclusion
To date, it is well admitted that Voc depends on the energy difference Δ(LUMOA–HOMOD). However, this value is the maximum theoretical value that can be reached. The difference between this theoretical value and the experimental Voc value is more or less large depending on the properties of the ED/EA interface such as the roughness of the bottom layer and charge recombination, some of them being incompressible [14] so that the value of Voc is well described by:
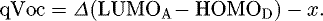 (1)
(1)
It was shown that, in the case of BHJ [15] the x value is at least 0.6 eV when the fullerene derivative, PCBM, is used as EA. In the case of PHJ, when C60 is used as EA, we have shown that, after optimization of the layer thicknesses, x is smaller than 0.6 eV. This makes that, by choosing ED and EA molecules with large Δ (LUMOA–HOMOD) value, OPVs with high efficiency could be achieved, even if the exciton charges separation is not as efficient as in the case of BHJ.
Acknowledgments
The authors would like to thank CNRST (PPR/2015/9) Ministry, Morocco for funding support.
References
- https://www.heliatek.com/en/ [Google Scholar]
- https://onlinelibrary.wiley.com/doi/full/10.1002/adem.201700 124 [Google Scholar]
- J.C. Bernède, J. Chil. Chem. Soc. 53 , 1549 (2008) [Google Scholar]
- K. Cnops, B.P. Rand, D. Cheyns, P. Heremans, Appl. Phys. Lett. 101 , 143301 (2012) [CrossRef] [Google Scholar]
- K. Cnops, B.P. Rand, D. Cheyns, B. Verreet, M.A. Empl, P. Heremans, Nat. Commun. 5 , 3406 (2014) [CrossRef] [PubMed] [Google Scholar]
- Z. El Jouad, M. Morsli, G. Louarn, L. Cattin, M. Addou, J.C. Bernède, Sol. Energy Mater. Sol. Cells 141 , 429 (2015) [CrossRef] [Google Scholar]
- M. Urbani, M-E. Ragoussi, M.K. Nazeeruddin, T. Torres, Coord. Chem. Rev. 381 , 1 (2019) [CrossRef] [Google Scholar]
- A. Mishra, P, Bäuerle, Angew. Chem. Int. Ed. 51 , 2020 (2012) [CrossRef] [Google Scholar]
- L. Cattin, S. Ouro Djobo, C. Sanchez, M. Morsli, J.C. Bernède, Energy Procedia 31 , 89 (2012) [CrossRef] [Google Scholar]
- E. Ortega, R. Montecinos, L. Cattin, F.R. Díaz, M.A. del Valle, J.C. Bernède, J. Mol. Struct. 114 , 615 (2017) [CrossRef] [Google Scholar]
- F. Martinez, G. Neculqueo, J.C. Bernède, L. Cattin, M. Makha, Phys. Status Solidi A 212 , 1767 (2015) [CrossRef] [Google Scholar]
- https://mgraham.com/gouache-paints/phthalocyanine-blue-140/ [Google Scholar]
- S.-W. Liu, W.-C. Su, C.-C. Lee, C.-C. Chou, C.-W. Cheng, ECS Solid State Lett. 1 , 70 (2012) [CrossRef] [Google Scholar]
- J. Benduhn, K. Tvingstedt, F. Piersimoni, S. Ullbrich, Y. Fan, M. Tropiano, K.A. McGarry, O. Zeika, M.K. Riede, C.J. Douglas, S. Barlow, S.R. Marder, D. Neher, D. Spoltore, K. Vandewal, Nat. Energy 2 , 17053 (2017) [CrossRef] [Google Scholar]
- D. Baran, T. Kirchartz, S. Wheeler, S. Dimitrov, M. Abdelsamie, J. Gorman, R.S. Ashraf, S. Holliday, A. Wadsworth, N. Gasparini, P. Kaienburg, H. Yan, A. Amassian, C.J. Brabec, J.R. Durrant, I. McCulloch, Energy Environ. Sci. 9 , 3561 (2016) [CrossRef] [Google Scholar]
Cite this article as: Zouhair El Jouad, Linda Cattin, Mohammed Addou, Jean Christian Bernède, Open circuit voltage of organic photovoltaic cells using C60 as acceptor: variation with the donor, Eur. Phys. J. Appl. Phys. 86, 20201 (2019)
All Tables
All Figures
 |
Fig. 1 Band structure of the interface donor/acceptor. |
| In the text | |
 |
Scheme 1 Commercial electron donors. |
| In the text | |
 |
Scheme 2 Laboratory made ED. |
| In the text | |
 |
Fig. 2 J–V characteristics of OPVs with Pentacene/ C60 and SubPc/C60 active couple. |
| In the text | |
 |
Fig. 3 Variation of (Δ(LUMOA–HOMOD) − Voc) with Δ(LUMOA–HOMOD) |
| In the text | |
 |
Fig. 4 3D image of a pentathiophene layer with thickness 70 nm. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


